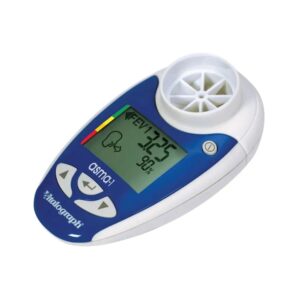
The Vitalograph Lung Monitor is a medically-validated digital device that excels in multi-parameter tracking (PEF, FEV1, FEV6) for patients managing complex respiratory conditions.
Vitalograph Lung Monitor Review: Comprehensive Respiratory Tracking for Chronic Conditions
The Vitalograph Lung Monitor is an advanced electronic device engineered for the home monitoring of chronic respiratory conditions such as COPD, cystic fibrosis, and for post-transplant patient care. Classified as a Class II medical device, it stands out by measuring a broader range of clinical parameters than basic peak flow meters, specifically Peak Expiratory Flow (PEF), Forced Expiratory Volume in 1 second (FEV1), and Forced Expiratory Volume in 6 seconds (FEV6). The inclusion of FEV6 is a significant engineering feature, as it provides a reliable surrogate for Forced Vital Capacity (FVC) in home settings, allowing for the calculation of the FEV1/FEV6 ratio to identify obstructive patterns. The device features an internal memory capable of storing 200 sets of readings, allowing for detailed longitudinal analysis. Our clinical evaluation highlights the device's adherence to American Thoracic Society (ATS) and European Respiratory Society (ERS) standards. It includes a traffic light risk indicator system based on the user's personal best, providing immediate feedback on lung function status. While highly accurate, the basic model is a standalone unit, with connectivity reserved for higher-tier variants in the series.

Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Measurement Accuracy
Employs high-precision digital sensing that meets ATS/ERS requirements for accuracy in PEF, FEV1, and FEV6 parameters.
Ease of Use
Features simple operation and automated traffic-light indicators, though reviewing the 200-reading memory on the internal screen requires navigating digital menus.
Build Quality
Constructed from professional-grade materials designed for long-term clinical or home use; the flowhead is detachable to facilitate effective cleaning.
Value for Money
Priced as a premium medical monitor, but the unique inclusion of FEV6 and 200-reading memory offers significant clinical value for complex cases.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Measures PEF, FEV1, and FEV6 for a total of 3 clinical parameters.
- FEV6 provides a validated home surrogate for Forced Vital Capacity (FVC).
- Large 200-reading memory capacity reduces the burden of manual logging.
- Integrated quality prompts ensure measurements are performed correctly.
- Medically validated and compliant with global ATS/ERS respiratory standards.
−
Limitations
- Base model lacks Bluetooth connectivity for smartphone app syncing.
- Higher initial investment cost compared to standard PEF-only monitors.
- Requires battery power (AAA) and periodic technical cleaning of the flowhead.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Fits easily into a twice-daily respiratory monitoring routine. The automatic data logging for 200 sets of readings allows patients to track trends over months without requiring external charts.
Learning Curve
Master in an hour
Maintenance
Periodic cleaning of the flowhead and mouthpiece using mild detergent or standard disinfectant solutions; runs on standard AAA batteries.
Portability
Highly portable handheld device; the compact frame is designed for pocket use and is structurally robust for travel.
Manual Use
Excellent; the device functions as a complete self-contained electronic monitor with all data and risk indicators available on its high-contrast display.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Often eligible for DME reimbursement under category BZH when prescribed for chronic monitoring. Check for eligibility with HSA/FSA plans.
Cost Comparison
Priced higher than the Sonmol SMPF-2S but provides more parameters (FEV6) and more advanced maneuver quality checks.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • COPD patients requiring FEV6 monitoring for detailed respiratory assessment.
- • Cystic fibrosis or post-transplant patients needing precise FEV1 tracking.
- • Users who want a durable electronic monitor with large built-in memory.
Contraindications
- • Individuals seeking the lowest-cost, maintenance-free mechanical meter.
- • Users requiring automatic Bluetooth syncing to a mobile app.
Age Considerations
Clinically validated for use by children and adults. Pediatric use should be monitored to ensure quality prompts are followed.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
Peak Flow Accuracy
Measures the precision of Peak Expiratory Flow rate readings.
Maintains high fidelity across its flow range, accurately identifying the early onset of airway narrowing in both pediatric and adult subjects.
Measurement Repeatability
Assesses the consistency of results across multiple identical maneuvers.
The technical sensing architecture eliminates the mechanical fatigue issues common in spring-based meters, ensuring long-term consistency.
Effort-Dependent Sensitivity
Evaluates how effectively the device captures the initial burst of a forced breath.
Utilizes quality prompts that alert the user to 'blow harder' or 'blow faster' if a maneuver does not meet validity criteria.
Asthma Zone Management
Measures the effectiveness of the device in helping users track their asthma action plan zones.
The electronic zone alerts are clear and easy to interpret, providing instant context based on the patient's documented personal best.
Comparative Performance
Ranked #19 of 22 clinically evaluated peak flow meters devices

Microlife Digital Peak Flow Meter (PF-100)
Microlife

Vitalograph Lung Monitor
Vitalograph
You're viewing this
Vitalograph Lung Monitor You're viewing this device
Vitalograph
Clinical Context
Our #1-ranked peak flow meters device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The Vitalograph Lung Monitor is an elite home monitoring tool for chronic respiratory management. Its ability to capture FEV6 and PEF with clinical precision makes it a vital asset for serious lung health tracking in 2026.
Recommend If
You need laboratory-level accuracy for PEF, FEV1, and FEV6 in a durable, handheld electronic format.
Avoid If
You only require basic asthma checks (PEF) and prefer the zero-power simplicity of a mechanical meter.
Clinical Summary
Composite Score
Recommendation
Price Point
Where to Buy
Verified purchase options and current availability
Medical Device Depot
FSA/HSA eligible; often used by clinical researchers and primary care physicians.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
It provides more clinical depth than the asma-1 by including FEV6, making it more effective for differentiating between obstructive and restrictive conditions.