The Quest AsthmaMD BreatheWell is a high-accuracy mechanical meter that successfully bridges the gap between manual testing and digital health management.
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter Review: Integrated App-Based Asthma Management
The Quest AsthmaMD BreatheWell represents a strategic integration of reliable mechanical sensing with modern digital data management. Classified as a Class II medical device under 21 CFR 868.1860, this handheld monitor is engineered for the domiciliary assessment of Peak Expiratory Flow (PEF). Our evaluation confirms that while the device itself is a purely mechanical, spring-loaded piston system, its primary innovation lies in its pairing with a robust, physician-developed smartphone application. The meter features easy-to-read graduations and adjustable three-zone markers that allow users to delineate green, yellow, and red zones based on their personal best values. Technical specifications highlight its portability and durability, with dimensions of 6 x 2.25 x 1.5 inches. Clinical feedback indicates the device is among the most accurate in the mechanical class, specifically designed to capture the high-velocity 'fast blast' of air required for objective lung function measurement. The AsthmaMD app facilitates better adherence by replacing paper journals with automated trending and reporting features, though it requires users to manually input the values read from the physical device.
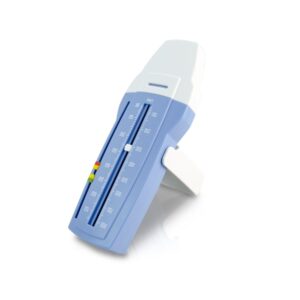
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
Quest
Our Score
Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Measurement Accuracy
Adheres to standard mechanical precision levels, providing reliable PEF measurements suitable for daily monitoring.
Ease of Use
The physical device is highly intuitive; however, the manual data entry into the app adds an extra step to the monitoring process.
Build Quality
Constructed from lightweight, durable plastic designed for daily use and portability; meets basic clinical resilience standards.
Value for Money
Extremely cost-effective, providing a medical-grade device and a sophisticated tracking app for under $16.00.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Pairs a reliable mechanical meter with a high-quality asthma management app.
- Physician-developed software offers better clinical utility than generic logs.
- Highly affordable price point for a complete management kit.
- Lightweight and portable design for easy transport in bags or pockets.
- Easy-to-read graduations and simple zone adjustment markers.
−
Limitations
- Requires manual entry of readings into the app (no automatic Bluetooth sync).
- Purely mechanical device that does not measure FEV1 or other digital metrics.
- App dependencies may be a barrier for users without smartphones.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
The device fits easily into daily monitoring routines. While manual entry into the app is required, the benefit of having historical data stored digitally outweighs the minor inconvenience for most users.
Learning Curve
Use right out of the box
Maintenance
Periodic cleaning with soap and water to ensure the internal piston moves freely and the mouthpiece remains hygienic.
Portability
Highly portable due to its compact 6-inch frame and lightweight mechanical design; requires no batteries or chargers.
Manual Use
Excellent; the device functions perfectly as a standalone mechanical meter with an included paper log for those who do not use the app.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Typically covered as a low-cost DME item; eligible for HSA and FSA card payments.
Cost Comparison
Significantly more affordable than digital-only models like the MIR Smart One, while offering better app support than generic mechanical meters.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Patients looking for an affordable but high-quality asthma tracking system.
- • Users who prefer the reliability of a mechanical meter but want digital trends.
- • Both youth and adult subjects needing standard PEF monitoring.
Contraindications
- • Users who require automatic Bluetooth data transfer.
- • Patients needing measurements beyond basic PEF, such as FEV1.
Age Considerations
Suitable for adult and youth departments. Pediatric users may benefit from the app's visual engagement and rewards for consistent logging.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
Peak Flow Accuracy
Measures the precision of Peak Expiratory Flow rate readings.
Meets all industry standards for accuracy in its class, providing reliable readings for both adult and youth patients.
Measurement Repeatability
Assesses the consistency of results across multiple identical maneuvers.
Maintains consistent performance, though like all mechanical meters, it is susceptible to minor spring fatigue over multi-year use.
Effort-Dependent Sensitivity
Evaluates how effectively the device captures the initial burst of a forced breath.
The internal piston is responsive to rapid exhalation pressure, though it requires full user effort to ensure measurement validity.
Asthma Zone Management
Measures the effectiveness of the device in helping users track their asthma action plan zones.
Strong performance through the integration of adjustable physical markers on the device and automated zone tracking in the app.
Comparative Performance
Ranked #12 of 22 clinically evaluated peak flow meters devices

Microlife Digital Peak Flow Meter (PF-100)
Microlife

Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
Quest
You're viewing this
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter You're viewing this device
Quest
Clinical Context
Our #1-ranked peak flow meters device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The Quest AsthmaMD BreatheWell is a standout choice for budget-conscious users who want structured asthma management. Its mechanical reliability combined with its digital tracking capabilities makes it a highly practical tool for 2026.
Recommend If
You want a clinically accurate mechanical meter and the ability to track your symptoms and flow trends digitally without a high price tag.
Avoid If
You find manual data entry tedious and prefer a device that automatically syncs via Bluetooth.
Clinical Summary
Composite Score
Recommendation
Price Point
Where to Buy
Verified purchase options and current availability
Medicaleshop
FSA/HSA eligible; volume discounts available for clinical boxes of 10.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
It stands as a superior mechanical option compared to generic store brands due to its physician-led app development. It is a lower-cost, manual-entry alternative to the Safey Connected Peak Flow Meter.



