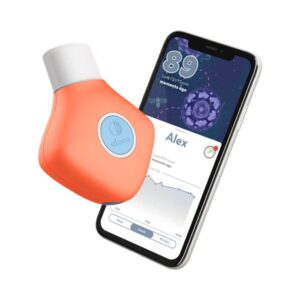
A robust, ATS/ERS 2019-compliant smart spirometer that pairs precise infrared turbine technology with a comprehensive app for seamless remote patient monitoring.
NuvoAir Air Next Review: Connected Spirometry for Scalable Remote Care
The NuvoAir Air Next is an advanced, FDA-cleared (K231416) smart spirometer designed to test lung function in adults and children aged 5 and older. Bridging the gap between clinic and home, it measures an extensive list of respiratory parameters including FVC, FEV1, and PEF. It utilizes a digital infrared interruption sensor to count the rotations of a propeller within a disposable turbine, converting these precise measurements into airflow data. Fully compliant with ATS/ERS 2019 technical standards, the device connects seamlessly via Bluetooth Low Energy to the NuvoAir companion app. The app provides real-time coaching and quality-of-blow feedback, ensuring patients perform maneuvers correctly. Its integration into broader 'Platform-as-a-Service' (PaaS) models makes the Air Next a highly scalable solution for healthcare providers managing chronic respiratory conditions remotely.

Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Measurement Accuracy
Achieves high accuracy (±3% for volume and ±5% for flow) using its infrared digital turbine sensor, fully meeting ATS/ERS 2019 compliance standards.
Ease of Use
The device is simple to use with disposable turbines that eliminate cleaning hassles, though proper FVC maneuvers still require patient effort.
App Quality
The app excels by offering real-time effort feedback and maneuver quality grading, ensuring patients generate usable clinical data.
Value for Money
Offers clinical-grade parameter tracking for complex disease management, though the required disposable turbines present an ongoing cost.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Measures over 30 respiratory parameters including full FVC, FEV1, and PEF.
- Fully compliant with ATS/ERS 2019 spirometry standards.
- Companion app provides real-time coaching and quality-of-blow feedback.
- Disposable turbines ensure maximum hygiene with zero cleaning required.
- Integrates seamlessly into clinical RPM and telehealth platforms.
−
Limitations
- Lacks a built-in display; totally dependent on a smartphone or tablet.
- Requires continuous purchasing of single-use disposable turbines.
- A full FVC maneuver can be challenging for patients without in-person coaching.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Requires keeping a smartphone handy and maintaining a supply of disposable turbines, but the automated syncing and in-app coaching make the testing process straightforward and reliable.
Learning Curve
Master in a day
Maintenance
Virtually zero cleaning maintenance is needed due to the single-use disposable turbines, though the user must ensure their smartphone is charged and connected.
Portability
Highly portable and easy to travel with, provided the user remembers to pack sufficient disposable turbines for their trip.
Manual Use
The device cannot be used manually; it has no screen and requires a Bluetooth connection to the NuvoAir app to operate and display results.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Typically covered when dispensed as part of a formal Remote Patient Monitoring (RPM) program prescribed by a pulmonologist or primary care physician.
Cost Comparison
The initial hardware is reasonably priced, but the recurring cost of single-use disposable turbines makes it more expensive to maintain than devices with reusable sensors.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Patients enrolled in provider-managed Remote Patient Monitoring programs.
- • Individuals managing complex conditions like severe asthma, COPD, or pulmonary fibrosis.
- • Users who prefer the hygiene of single-use turbines over cleaning reusable components.
Contraindications
- • Users looking for a standalone device with a built-in screen.
- • Patients who want to avoid the recurring cost of disposable supplies.
Age Considerations
FDA-cleared for use in adults and children 5 years of age and older, with the app providing necessary coaching for pediatric compliance.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
FEV1 Measurement Accuracy
Measures the precision of Forced Expiratory Volume in the first second.
Maintains highly precise FEV1 tracking that correlates directly with clinical laboratory findings.
FVC Measurement Accuracy
Measures the precision of Forced Vital Capacity readings.
Accurately captures total exhaled volume with real-time feedback, calculating critical metrics like the FEV1/FVC ratio.
Longitudinal Trend Tracking
Evaluates the device's ability to consistently track respiratory changes over time.
Synchronizes data securely via the app, making it an excellent tool for logging daily trends over long monitoring periods.
Clinical Spirometry Correlation
Assesses how closely the home measurements align with formal clinical pulmonary function tests.
The digital infrared interruption sensor provides data highly concordant with in-clinic spirometry, validating its use in remote care.
Comparative Performance
Ranked #5 of 14 clinically evaluated home spirometers devices

MIR Spirobank Oxi
MIR
Aluna 2 Smart Spirometer
Aluna

NuvoAir Air Next
NuvoAir
You're viewing this
NuvoAir Air Next You're viewing this device
NuvoAir
Clinical Context
Our #1-ranked home spirometers device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The NuvoAir Air Next is an excellent, clinic-grade smart spirometer that excels in structured remote patient monitoring environments. Its reliance on disposable turbines minimizes maintenance while its powerful app ensures patients perform valid, actionable tests at home.
Recommend If
You need comprehensive respiratory tracking (FVC, FEV1) with real-time app coaching to ensure you are performing tests correctly, and you prefer the hygiene of disposable turbines.
Avoid If
You want to avoid ongoing consumable costs associated with disposable turbines, or if you prefer a device with an onboard display.
Clinical Summary
Composite Score
Recommendation
Where to Buy
Verified purchase options and current availability
NuvoAir Official Store
FSA/HSA eligible; volume pricing available for clinical providers.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
It rivals the MIR Spirobank Smart in terms of offering a comprehensive parameter list and clinical-grade full-loop testing. However, the NuvoAir ecosystem leans heavily into disposable turbines for hygiene, whereas MIR offers reusable options.