Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter vs Safey Connected Peak Flow Meter - Clinical Comparison
Compare Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter and Safey Connected Peak Flow Meter side-by-side with clinical performance scores, FDA status, pricing, and expert recommendations
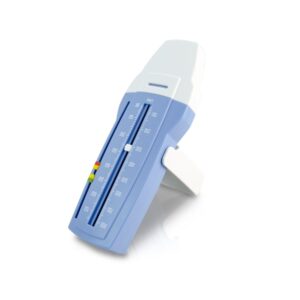
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
Quest

Performance Metrics
Performance Metrics
Measurement Accuracy
Measures the closeness of the device's readings to a known reference standard.
Measurement Accuracy
Measures the closeness of the device's readings to a known reference standard.
Ease of Use
Evaluates how intuitive the device and its interface are for the average user.
Ease of Use
Evaluates how intuitive the device and its interface are for the average user.
Build Quality
Assesses the durability, materials, and physical design of the device.
Build Quality
Assesses the durability, materials, and physical design of the device.
Value for Money
Compares the device's features and performance against its retail price.
Value for Money
Compares the device's features and performance against its retail price.
Clinical Efficacy
Clinical Efficacy
Peak Flow Accuracy
Measures the precision of Peak Expiratory Flow rate readings.
Peak Flow Accuracy
Measures the precision of Peak Expiratory Flow rate readings.
Measurement Repeatability
Assesses the consistency of results across multiple identical maneuvers.
Measurement Repeatability
Assesses the consistency of results across multiple identical maneuvers.
Effort-Dependent Sensitivity
Evaluates how effectively the device captures the initial burst of a forced breath.
Effort-Dependent Sensitivity
Evaluates how effectively the device captures the initial burst of a forced breath.
Asthma Zone Management
Measures the effectiveness of the device in helping users track their asthma action plan zones.
Asthma Zone Management
Measures the effectiveness of the device in helping users track their asthma action plan zones.
Specifications
| Specification | Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter | Safey Connected Peak Flow Meter |
|---|---|---|
| Price | $20 | $99 |
| FDA Status | FDA 510(k) Cleared | FDA 510(k) Cleared |
| Clinical Evidence | Moderate Clinical Evidence | Strong Clinical Evidence |
| Clinical Recommendation | Recommended | Highly Recommended |
| Connectivity | N/A |
bluetooth
|
Value for Money
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
Strengths
- ✓ Pairs a reliable mechanical meter with a high-quality asthma management app.
- ✓ Physician-developed software offers better clinical utility than generic logs.
- ✓ Highly affordable price point for a complete management kit.
- ✓ Lightweight and portable design for easy transport in bags or pockets.
- ✓ Easy-to-read graduations and simple zone adjustment markers.
Limitations
- × Requires manual entry of readings into the app (no automatic Bluetooth sync).
- × Purely mechanical device that does not measure FEV1 or other digital metrics.
- × App dependencies may be a barrier for users without smartphones.
Safey Connected Peak Flow Meter
Strengths
- ✓ Measures both PEF and FEV1 for comprehensive lung health tracking.
- ✓ High-precision infrared turbine sensor reduces mechanical error.
- ✓ Seamless Bluetooth connectivity for automatic data logging.
- ✓ Intuitive traffic light risk indicator system within the app.
- ✓ Portable, pocket-sized design suitable for travel and home use.
Limitations
- × Higher upfront cost compared to basic mechanical meters.
- × Requires a compatible smartphone for full functionality and data viewing.
- × Initial Bluetooth pairing and app setup may be difficult for some elderly users.
Expert Review Summary
Our clinical assessment and verdict for each device
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
"The Quest AsthmaMD BreatheWell is a high-accuracy mechanical meter that successfully bridges the gap between manual testing and digital health management."
The Quest AsthmaMD BreatheWell represents a strategic integration of reliable mechanical sensing with modern digital data management. Classified as a Class II medical device under 21 CFR 868.1860, this handheld monitor is engineered for the domiciliary assessment of Peak Expiratory Flow (PEF). Our evaluation confirms that while the device itself is a purely mechanical, spring-loaded piston system, its primary innovation lies in its pairing with a robust, physician-developed smartphone application. The meter features easy-to-read graduations and adjustable three-zone markers that allow users to delineate green, yellow, and red zones based on their personal best values. Technical specifications highlight its portability and durability, with dimensions of 6 x 2.25 x 1.5 inches. Clinical feedback indicates the device is among the most accurate in the mechanical class, specifically designed to capture the high-velocity 'fast blast' of air required for objective lung function measurement. The AsthmaMD app facilitates better adherence by replacing paper journals with automated trending and reporting features, though it requires users to manually input the values read from the physical device.
Safey Connected Peak Flow Meter
"The Safey Connected Peak Flow Meter is a top-tier digital respiratory monitor that excels in accuracy and data integration for patients requiring detailed lung health tracking."
The Safey Connected Peak Flow Meter represents a significant advancement over traditional mechanical devices by integrating a high-precision turbine sensor with mobile connectivity. As an FDA-cleared Class II medical device, it measures both Peak Expiratory Flow (PEF) and Forced Expiratory Volume in one second (FEV1), providing a more comprehensive view of lung function than standard meters. Our evaluation focused on the device's technical architecture, specifically its use of the infrared interrupt concept. This design uses a vane that rotates based on airflow, with infrared pairs detecting rotation speed and direction to calculate results. This method significantly reduces the friction-related errors often seen in mechanical spring-based systems. Clinical performance data demonstrates that the device adheres strictly to American Thoracic Society (ATS) and European Respiratory Society (ERS) standards for spirometry. The companion mobile application automatically categorizes results into standardized green, yellow, and red zones, simplifying the implementation of asthma action plans. The build quality is robust, featuring medical-grade polymers and an IP22 ingress protection rating, making it suitable for both home and clinical environments.
Where to Buy
Verified purchase options and current availability
Quest AsthmaMD BreatheWell Lung Performance Peak Flow Meter
Medicaleshop
$17.01Safey Connected Peak Flow Meter
Top ChoiceWalmart
$98.99Zone Medical
$199.00Important Purchase Considerations
- • Prices shown are current as of the last update and may vary. Always verify pricing at the retailer's website.
- • Some medical devices may require a prescription. Check with your healthcare provider before purchasing.
- • Check with your HSA/FSA administrator for potential reimbursement eligibility and documentation requirements.
- • Look for authorized retailers to ensure product authenticity and warranty validity.