A highly engaging, hospital-grade smart spirometer that utilizes gamification to encourage daily testing adherence in pediatric and adult patients.
Aluna 2 Smart Spirometer Review: Gamified Respiratory Tracking for Asthma and CF
The Aluna 2 is an innovative over-the-counter and prescription smart spirometer intended for monitoring FEV1 (Forced Expiratory Volume in one second) and PEF (Peak Expiratory Flow). Unlike many competitors that use mechanical turbines, the Aluna 2 utilizes a Lilly type pneumotachometer with a differential pressure sensor across a stainless-steel mesh to calculate airflow and volume, offering exceptional durability and hospital-grade accuracy. Cleared by the FDA (K232588) and fully compliant with ATS/ERS 2019 standards, it communicates via Bluetooth 4.0 LE to a dedicated mobile app available on iOS and Android. The standout feature of the Aluna 2 ecosystem is its highly developed companion app, which incorporates engaging breathing games that can be toggled on or off, making it particularly effective for boosting daily adherence in children and teens managing asthma or cystic fibrosis.
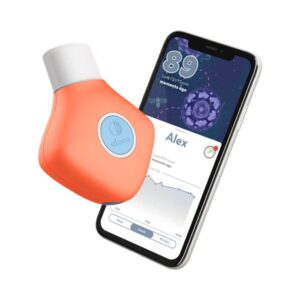
Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Measurement Accuracy
The differential pressure sensor delivers hospital-grade precision with FEV1 linearity of <2.5%, easily meeting the rigorous ATS/ERS 2019 technical updates.
Ease of Use
Extremely user-friendly. The rechargeable battery and simple blow-through design make it highly accessible, especially for children.
App Quality
The companion app is top-tier, featuring seamless cloud syncing, data export tools, and engaging breathing games that can be customized to the user's preference.
Value for Money
Offers immense value for families struggling with testing adherence, though it lacks the FVC diagnostic capabilities found in similarly priced clinical models.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Highly engaging gamified app significantly improves testing adherence.
- Uses a durable Lilly type pneumotachometer with a differential pressure sensor.
- Built-in rechargeable battery eliminates the need for constant battery replacements.
- FDA-cleared and fully compliant with ATS/ERS 2019 standards.
- Games can be toggled off for adult users who prefer a standard interface.
−
Limitations
- Does not measure Forced Vital Capacity (FVC).
- Relies entirely on a paired smartphone or tablet to display readings.
- Requires periodic charging via USB.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Transforms daily respiratory testing from a medical chore into a fun, interactive game, making it exceptionally easy to integrate into a child's morning or evening routine.
Learning Curve
Use right out of the box
Maintenance
Requires regular charging of the internal battery and periodic cleaning of the device airway to ensure the stainless-steel mesh pressure sensor remains unobstructed.
Portability
Highly portable and travel-friendly. The compact design and rechargeable battery make it easy to pack and use on the go.
Manual Use
Cannot be used manually; the device lacks a built-in display and relies entirely on the Bluetooth connection to the Aluna mobile app to function.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Frequently covered when distributed through clinical Remote Patient Monitoring (RPM) programs, or it can be purchased using FSA/HSA funds.
Cost Comparison
Competitively priced within the smart spirometer market, offering distinct value for families due to the sophisticated software and pressure-sensor hardware.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Children (aged 5+) and adolescents managing asthma or cystic fibrosis.
- • Patients who struggle with adherence to daily monitoring regimens.
- • Clinics looking for a patient-friendly device for RPM programs.
Contraindications
- • Patients with complex COPD requiring full FVC loop measurements.
- • Individuals who do not own a compatible smartphone (iOS 15+ or Android 8+).
Age Considerations
Designed for pediatric to adult users, but specifically noted by the FDA as not recommended for children under 5 years of age.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
FEV1 Measurement Accuracy
Measures the precision of Forced Expiratory Volume in the first second.
Demonstrates excellent accuracy with an FEV1 repeatability of ±2.5% or 0.05 L (whichever is greater), tracking airway obstruction with clinical reliability.
Longitudinal Trend Tracking
Evaluates the device's ability to consistently track respiratory changes over time.
The app securely transmits data to a cloud server via an API, maintaining an impeccable longitudinal record of symptoms and lung function.
Clinical Spirometry Correlation
Assesses how closely the home measurements align with formal clinical pulmonary function tests.
The Lilly type pneumotachometer closely correlates with in-office spirometry for FEV1 and PEF, making the data highly actionable for clinicians.
Comparative Performance
Ranked #2 of 14 clinically evaluated home spirometers devices

MIR Spirobank Oxi
MIR
Aluna 2 Smart Spirometer
Aluna

Aluna 2 Smart Spirometer Current
Aluna
Clinical Context
This device scored 1 points behind the highest-rated device in this category. Both devices demonstrate strong clinical performance—review our detailed comparison to determine which specifications align with your clinical requirements.
Clinical Recommendation
Final assessment and prescribing guidance
The Aluna 2 is an outstanding smart spirometer that successfully tackles the major clinical challenge of patient adherence. By pairing hospital-grade pressure sensor accuracy with a genuinely engaging gamified app, it is a top recommendation for children and adults managing asthma or cystic fibrosis.
Recommend If
You or your child struggle with adherence to daily lung monitoring and would benefit from an interactive, gamified experience that rewards consistency.
Avoid If
You require full FVC loop measurements for advanced disease management, or if you prefer a standalone device with a built-in screen.
Clinical Summary
Composite Score
Recommendation
Where to Buy
Verified purchase options and current availability
Aluna Store
FSA/HSA eligible; requires prescription for some insurance billing.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
Similar to the CMI Health SpiroLink, the Aluna 2 utilizes a pressure sensor rather than the mechanical turbine found in the MIR Smart One. However, the Aluna 2 stands entirely apart with its heavily gamified app experience tailored for adherence.


