The Nascool Digital Peak Flow Meter is a highly accessible standalone device that delivers accurate digital lung function metrics without the complexity of app connectivity.
Nascool Digital Peak Flow Meter Review: Affordable and Precise Standalone Monitoring
The Nascool Digital Peak Flow Meter (Model SMPF-2S) is an FDA-cleared Class II medical device designed for home monitoring of asthma and other respiratory conditions. Manufactured by Shanghai Sonmol Medical Equipment Co., Ltd., this device utilizes a pressure sensor sensing modality to measure both Peak Expiratory Flow (PEF) and Forced Expiratory Volume in one second (FEV1). Technical specifications confirm that the device meets American Thoracic Society (ATS) 2005 standards for accuracy and precision. The unit features an integrated LCD screen that provides immediate digital feedback, including a traffic-light risk indicator system (Green, Yellow, Red) based on the user's personal best. Unlike Bluetooth-enabled models, this iteration is designed for manual record-keeping, prioritizing standalone utility and long-term battery life. The hardware is constructed from medical-grade ABS plastic with an unremovable mouthpiece, which has been validated for single-user reuse. Clinical performance testing against certified pulmonary waveforms demonstrates high fidelity in capturing airway obstruction trends in both pediatric and adult subjects.
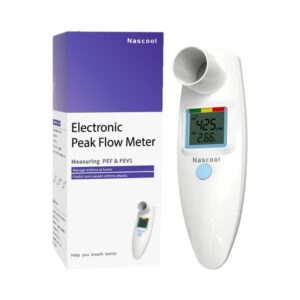
Nascool Digital Peak Flow Meter (Manual Recording)
Nascool
Our Score
Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Measurement Accuracy
Pressure sensor architecture provides accuracy within ±10% for PEF and ±3% for FEV1, meeting or exceeding standard requirements for home clinical monitors.
Ease of Use
The standalone LCD design is highly intuitive, providing immediate results with zero setup or smartphone pairing required.
Build Quality
Compact and lightweight construction using medical-grade ABS plastic; the unremovable mouthpiece is sturdy but requires thorough technical cleaning.
Value for Money
One of the most affordable digital meters capable of measuring FEV1, offering exceptional value for patients needing precise electronic monitoring on a budget.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Measures both PEF and FEV1 for comprehensive lung function tracking.
- Highly affordable price point for a medical-grade digital meter.
- Standalone operation requires no smartphone or Bluetooth pairing.
- Large, clear LCD display with integrated risk indicator zones.
- Compact and lightweight design (52g) for high portability.
−
Limitations
- Does not feature internal data memory or Bluetooth data logging.
- Requires manual record-keeping in a paper diary for trend analysis.
- Mouthpiece is unremovable, making cleaning slightly more involved.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
The device is quick and efficient for daily morning and evening checks. Because it is battery-operated and standalone, it can be kept on a bedside table for immediate use without needing to launch a mobile application.
Learning Curve
Use right out of the box
Maintenance
Periodic cleaning with soap and water; the unremovable mouthpiece should be rinsed carefully and fully air-dried. Operates on 2 AAA batteries.
Portability
Highly portable; its small dimensions (142x48x56mm) and lightweight frame make it a resilient travel monitor that does not require charging cables.
Manual Use
Outstanding; the device is specifically engineered for manual users, providing large fonts and clear color indicators on the built-in screen.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Often eligible for HSA/FSA reimbursement. Users should verify DME coverage for BZH-classified monitors with their specific provider.
Cost Comparison
Significantly more affordable than high-end Bluetooth spirometers like the MIR Smart One, while offering higher precision and more parameters than basic mechanical meters.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Budget-conscious patients who prefer standalone digital precision.
- • Users who want to monitor FEV1 without using a smartphone app.
- • Families needing an easy-to-read device for both kids and adults.
Contraindications
- • Individuals who require automated digital trending and cloud data storage.
- • Patients with severe obstruction needing measurements below 60 L/min.
Age Considerations
Clinically designed for adults and children over 5 years of age. Pediatric subjects require adult supervision to ensure correct technique.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
Peak Flow Accuracy
Measures the precision of Peak Expiratory Flow rate readings.
Reliably measures flow within a range of 60L/min to 840L/min, providing suitable range for both pediatric and adult subjects.
Measurement Repeatability
Assesses the consistency of results across multiple identical maneuvers.
Maintains consistent performance during multiple maneuvers, though digital pressure sensors require regular mouthpiece hygiene to prevent sensing drift.
Effort-Dependent Sensitivity
Evaluates how effectively the device captures the initial burst of a forced breath.
Pressure-based sensing is highly responsive to the initial exhalation spike, accurately identifying peak flow velocities.
Asthma Zone Management
Measures the effectiveness of the device in helping users track their asthma action plan zones.
Excellent visual feedback via an on-screen traffic light indicator that categorizes measurements relative to the user's personal best baseline.
Comparative Performance
Ranked #7 of 22 clinically evaluated peak flow meters devices

Microlife Digital Peak Flow Meter (PF-100)
Microlife

Nascool Digital Peak Flow Meter (Manual Recording)
Nascool
You're viewing this
Nascool Digital Peak Flow Meter (Manual Recording) You're viewing this device
Nascool
Clinical Context
Our #1-ranked peak flow meters device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The Nascool Digital Peak Flow Meter is an excellent choice for straightforward lung function tracking. Its ability to measure FEV1 and provide visual risk zones on a standalone screen makes it a high-value clinical tool for 2026.
Recommend If
You want an accurate digital monitor for PEF and FEV1 that is easy to read and very affordable, and you don't mind manually recording your readings.
Avoid If
You want your respiratory data to automatically sync to a smartphone or you require internal device memory for trend review.
Clinical Summary
Composite Score
Recommendation
Price Point
Where to Buy
Verified purchase options and current availability
Walmart
FSA/HSA eligible; price for manual model without Bluetooth.
Nascool Store
Manufacturer direct shipping.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
It provides the same core technical accuracy as the Sonmol Bluetooth model but trades connectivity for a lower price. It is more portable than the Microlife PF-100 but lacks its internal memory.



