A high-precision, FDA-cleared analytical platform that transforms wearable cardiac and oximetry data into hospital-grade sleep diagnostic reports.
SleepImage System (SaMD) Review: Medically-Validated Cloud-Based Sleep Diagnostics
The SleepImage System is a cloud-based Software as a Medical Device (SaMD) intended to aid healthcare professionals in the evaluation, diagnosis, and management of sleep disorders. Unlike basic lifestyle trackers, this system is FDA-cleared to establish sleep quality and provide a SleepImage Apnea Hypopnea Index (sAHI) for children, adolescents, and adults. The platform operates by automatically analyzing digital signals-typically Electrocardiogram (ECG) or Plethysmogram (PLETH) paired with oxygen saturation (SpO2)-collected during sleep via Bluetooth-compatible data sources like the SleepImage PO6 ring or standardized EDF files. Clinical evaluation of the system is robust, involving the analysis of over 2,000 sleep studies to confirm that its auto-scoring algorithms generate outputs comparable to manual human scoring from polysomnography (PSG). The software provides detailed graphical and numerical reports on sleep latency, duration, quality, and pathology, accessible through any major web browser.
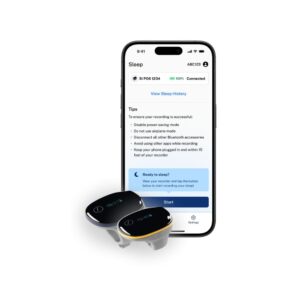
Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Diagnostic Accuracy
Demonstrated agreement levels with manually scored AHI from PSG studies sufficient to aid clinical diagnosis in both pediatric and adult populations.
Ease of Use
Automatic report generation and cloud-based access are highly convenient, though it requires a prescription and professional interpretation.
Wearing Comfort
As a software system, comfort depends on the chosen hardware; the compatible PO6 ring is a low-profile, non-intrusive wearable.
Value for Money
High clinical value given the FDA clearance for pediatric diagnostics and multi-modal heart/sleep analytics.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- FDA-cleared for diagnosis aid of sleep-disordered breathing in children and adults.
- Cloud-based system provides seamless automatic processing and report generation.
- Highly versatile; accepts PLETH, ECG, and SpO2 data from various hardware sources.
- Validated on a massive clinical dataset of over 2,000 subjects.
- Enables professional manual scoring and editing of auto-detected events.
−
Limitations
- Restricted to prescription use only by licensed healthcare practitioners.
- Does not issue real-time alarms and is not intended for active patient monitoring.
- Requires a separate Bluetooth-compatible data source (hardware) for signal acquisition.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Passive overnight data collection via wearable hardware, followed by automatic data upload and processing upon waking; clinicians can then review reports at any time via web portal.
Learning Curve
Master in an hour
Maintenance
Periodic firmware updates for compatible hardware; cloud software is maintained and updated automatically by the manufacturer.
Portability
High; the system is cloud-based and accessible from any internet-connected computing platform.
Manual Use
N/A (Software-based system requiring a computing platform for any interaction)[cite: 530, 549].
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
As a prescription diagnostic tool, it is often covered when part of a clinical evaluation for sleep apnea management.
Cost Comparison
Cost is primarily associated with the required hardware and clinician's service fees rather than a standalone software purchase.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Pediatric patients requiring medically cleared home sleep diagnostics.
- • Clinicians needing a scalable, cloud-based platform for remote sleep apnea management.
- • Users seeking clinical-grade sleep quality metrics validated against PSG.
Contraindications
- • Individuals seeking an over-the-counter (OTC) diagnostic solution without doctor involvement.
- • Patients needing real-time monitoring with audible alarms for critical events.
Age Considerations
Medically cleared for use in children, adolescents, and adults.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
AHI Detection Accuracy
Measures accuracy in identifying Apnea-Hypopnea Index levels.
Clinical evaluations confirmed that the sAHI auto-scoring algorithm generates outputs comparable to human manual scoring of PSG studies.
Sleep Stage Identification
Assesses the accuracy of identifying REM, Light, and Deep sleep states.
Provides analysis of sleep latency, duration, and pathology based on peer-reviewed guidelines from the AASM.
Respiratory Event Detection
Evaluates accuracy in detecting individual breathing disturbances.
Automatically calculates respiratory events by analyzing desaturations from SpO2 and coupling heart rate variability with respiration.
In-Lab PSG Correlation
Measures how the device results correlate with in-lab polysomnography.
Confirmed comparable outputs for PLETH vs ECG signals compared to standard PSG across more than 2,000 subjects.
Comparative Performance
Ranked #9 of 25 clinically evaluated home sleep tests devices

Withings Sleep Analyzer
Withings

Sunrise Home Sleep Test
Sunrise SA
SleepImage System (SaMD)
SleepImage
You're viewing this
SleepImage System (SaMD) You're viewing this device
SleepImage
Clinical Context
Our #1-ranked home sleep tests device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The SleepImage System (SaMD) is a premier clinical tool for home-based sleep apnea diagnostics. Its unique clearance for pediatric use and its ability to turn data from simple wearables into high-fidelity diagnostic reports make it a cornerstone of modern remote sleep medicine.
Recommend If
You need medically-validated sleep apnea screening or clinical sleep quality reports that are shareable with a healthcare provider.
Avoid If
You want an all-in-one lifestyle tracker that does not require a prescription or clinical oversight.
Clinical Summary
Composite Score
Recommendation
Alternative Devices
Offers significantly more clinical diagnostic utility and pediatric clearance than general wellness wearables like the Oura Ring or WHOOP.


