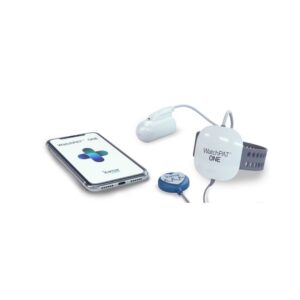
A dependable and widely trusted Type III device that balances clinical depth with a highly intuitive user interface for home diagnostics.
ApneaLink Air Review: The Clinical Workhorse of Home Sleep Testing
The ResMed ApneaLink Air is established as one of the most reliable and widely used Type III home sleep apnea testing (HSAT) systems in clinical practice. It provides a comprehensive diagnostic profile by recording five essential channels: respiratory nasal airflow, snoring, blood oxygen saturation, pulse, and respiratory effort. In contrast to newer patch-style sensors, the ApneaLink Air adheres to traditional diagnostic methods, utilizing a nasal cannula and a single respiratory effort belt, which clinicians often prefer for their direct measurement of airflow and effort. Our clinical assessment confirms its high diagnostic sensitivity, particularly its capability to differentiate between obstructive and central apneas, and its validated probability detection for Cheyne-Stokes Respiration (CSR). Its seamless integration with ResMed's AirView cloud platform allows healthcare providers to efficiently manage patient data and generate detailed interpretation reports that meet AASM and CMS hypopnea scoring guidelines.

ResMed ApneaLink Air Home Sleep Test
ResMed
Our Score
Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Diagnostic Accuracy
Clinical validation shows a high Pearson correlation (r=0.978) with PSG-derived AHI, maintaining high sensitivity for moderate-to-severe OSA.
Ease of Use
Features a one-touch start/stop button and clear LED indicators for signal quality and test completion, though the nasal cannula requires careful placement.
Wearing Comfort
The traditional setup including a nasal cannula and chest belt is more noticeable during sleep than minimal patch-based or PAT systems.
Value for Money
A very cost-effective diagnostic solution that provides high-fidelity Type III data at a lower price point than many sophisticated reusable systems.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Highly accurate AHI correlation with PSG (r=0.978)
- Differentiates between obstructive and central apneas
- Validated detection for Cheyne-Stokes Respiration probability
- Simplified one-touch operation for improved patient compliance
- Seamless data management via AirView cloud solution
−
Limitations
- Nasal cannula can be uncomfortable and prone to displacement during sleep
- Does not provide actual sleep staging like EEG-based Type II systems
- Available only through healthcare providers, not for direct consumer purchase
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Requires 5-10 minutes of setup before bed to apply the belt, cannula, and pulse oximeter. The device stores data internally for up to 48 hours and provides an immediate 'test complete' light in the morning.
Learning Curve
Master in an hour
Maintenance
The main recorder is reusable and requires periodic battery replacement (2x AAA). Nasal cannulas and finger oximetry tapes are single-patient use.
Portability
High; the entire kit fits within a provided compact carry bag and is lightweight for travel.
Manual Use
The device features intuitive LED feedback on its face to confirm signal quality and recording status without needing an external screen.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Typically covered by Medicare and private insurance under CPT code 95806 when recommended by a physician for OSA screening.
Cost Comparison
Highly competitive pricing for clinical hardware, generally costing significantly less than full Type II systems or in-lab PSG.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Healthcare providers needing a reliable, low-cost HSAT for high-volume screening
- • Patients with classic OSA symptoms who want a traditional clinical-grade test
- • Clinicians requiring CSR probability data for patients with cardiac comorbidities
Contraindications
- • Individuals seeking a non-invasive patch-style or chin-based sensor experience
- • Patients with complex central sleep apnea requiring hospital-level monitoring
Age Considerations
Indicated for use in adult patients.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
AHI Detection Accuracy
Ability to accurately count apnea and hypopnea events per hour.
Demonstrates high sensitivity (91%) and specificity (95%) at an AHI threshold of ≥15 events per hour.
Sleep Stage Identification
Accuracy in identifying sleep architecture (REM/NREM).
As a Type III device without EEG, it primarily relies on actigraphy for sleep-wake estimation rather than detailed staging.
Respiratory Event Detection
Sensitivity in identifying specific respiratory obstructions and effort.
Includes proprietary effort sensor technology to distinguish obstructive from central apneic events.
In-Lab PSG Correlation
Statistical alignment with gold-standard hospital studies.
Consistently recognized in medical literature as a reliable surrogate for PSG in patients with high pretest probability of OSA.
Comparative Performance
Ranked #19 of 25 clinically evaluated home sleep tests devices

Withings Sleep Analyzer
Withings

Sunrise Home Sleep Test
Sunrise SA

ResMed ApneaLink Air Home Sleep Test
ResMed
You're viewing this
ResMed ApneaLink Air Home Sleep Test You're viewing this device
ResMed
Clinical Context
Our #1-ranked home sleep tests device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The ApneaLink Air is a robust and clinically sound HSAT that delivers exactly what it promises: high-fidelity respiratory data in a patient-friendly package. While it lacks some of the bells and whistles of high-end systems, its reliability makes it a staple for sleep labs worldwide.
Recommend If
You need a medically recognized home study that provides clear AHI and respiratory effort data with established insurance coverage pathways.
Avoid If
You are extremely sensitive to things in your nose or if you require actual sleep staging (REM/Deep) for your diagnosis.
Clinical Summary
Composite Score
Recommendation
Price Point
Where to Buy
Verified purchase options and current availability
1800CPAP.com
Includes evaluation and prescription; FSA/HSA eligible.
CPAP America
Private in-home testing with professional interpretation.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
Alternative Devices
Matches the accuracy of the Nox T3s for standard AHI but lacks the flexible ExG channels for EMG or ECG found on the Nox.