A powerhouse of diagnostic depth, providing calibrated respiratory effort and optional ExG channels for clinicians who require more than just AHI data.
Nox T3s Review: The Standard for Sophisticated Type III Home Sleep Testing
The Nox T3s from Nox Medical is widely considered the clinical workhorse of Type III home sleep apnea testing (HSAT). Unlike many simplified home tests, the T3s utilizes sophisticated Respiratory Inductance Plethysmography (RIP) through its patented Nox RIP belts, which provide a calibrated flow signal (Nox Flow) on par with hospital-grade pneumotachography. This allows for highly reliable respiratory effort measurements even if the nasal cannula becomes dislodged. A major differentiator for the T3s is its two customizable bipolar ExG channels, which can be configured to record ECG, EMG, or even limited EEG for more complex diagnostic needs. The system also features Nox BodySleep™, an AI-powered algorithm that estimates sleep stages and wakefulness by analyzing respiratory patterns without requiring head-mounted sensors. Our evaluation highlights its exceptional signal quality and its pediatric-ready design, being cleared for patients as young as 2 years old.

Table of Contents
Clinical Summary
Clinical Performance Metrics
Quantitative assessment based on clinical testing protocols
Diagnostic Accuracy
Demonstrates a Pearson correlation of 0.93 with PSG-derived AHI and high sensitivity (92%) for moderate-to-severe OSA.
Ease of Use
Requires more preparation than simplified sensors, including applying two chest/abdominal belts and a nasal cannula, which may be challenging for some users.
Wearing Comfort
While ergonomic, the multi-sensor setup (belts, oximeter, and cannula) is more intrusive than minimal-contact chin or finger sensors.
Value for Money
Offers exceptional diagnostic depth and reusability, though it typically involves higher logistical costs for clinics compared to disposable units.
Quick Take
Key findings from our clinical evaluation
+
Strengths
- Calibrated RIP technology (Nox Flow) acts as a reliable backup to cannula flow.
- Customizable bipolar channels allow for ECG, EMG, or EEG recording.
- Pediatric-ready for children 2 years and older.
- AI-powered BodySleep™ estimates TST and sleep stages without EEG.
- Extremely low failure rates due to durable hardware and signal redundancy.
−
Limitations
- Complex multi-sensor setup may lead to user error without clear instruction.
- Nasal cannula and dual belts are less comfortable than minimalist 'patch' tests.
- Requires PC software (Noxturnal) for data download and detailed manual scoring.
Real-World Usage
Practical considerations for daily operation
Daily Routine Impact
Requires 10-15 minutes of setup time before bed to apply belts, cannula, and oximeter. Users must follow a specific 'hook-up' sequence. The device records locally and must be returned to the clinic for data retrieval.
Learning Curve
Master in an hour
Maintenance
Belts and cannula are typically single-patient use; the recorder itself requires periodic cleaning and battery replacement (1x AA).
Portability
High; the kit includes a dedicated carry case designed for hygiene and easy transport.
Manual Use
The device has an onboard OLED display for status indication and signal integrity checks, but detailed results require software analysis.
Cost & Coverage Analysis
Financial considerations and HSA / FSA eligibility
Pricing Breakdown
HSA/FSA Guidance
Standard Type III HSAT often covered under CPT 95806; clinicians can justify medical necessity for patients requiring effort-belt diagnostics.
Cost Comparison
Hardware cost is moderate, but clinical implementation usually involves a service model (HSTaaS) with predictable budgeting.
Patient Suitability
Clinical indications and contraindications
Indicated For
- • Clinicians needing high-fidelity respiratory effort data for complex cases.
- • Patients requiring bruxism or PLM analysis in addition to sleep apnea.
- • Pediatric sleep testing where calibrated RIP is essential.
Contraindications
- • Patients seeking a 'one-and-done' disposable patch-style experience.
- • Individuals with severe cognitive impairments who cannot manage the multi-belt setup.
Age Considerations
FDA cleared for patients greater than 2 years of age.
Clinical Efficacy Assessment
Evidence-based evaluation of clinical performance
AHI Detection Accuracy
Ability to accurately count apnea and hypopnea events.
Maintains high sensitivity even in mild cases; clinical studies show 92.5% accuracy for AHI ≥ 5.
Sleep Stage Identification
Accuracy in identifying REM and NREM states.
Nox BodySleep AI differentiates REM, NREM, and Wake with a Cohen's Kappa of 0.61-0.80, a substantial reliability for an EEG-free method.
Respiratory Event Detection
Sensitivity in identifying specific arousals and effort levels.
Calibrated dual-belt RIP provides true respiratory effort signals, essential for distinguishing central from obstructive apnea.
In-Lab PSG Correlation
Statistical alignment with in-hospital studies.
Widely recognized as meeting AASM standards for home testing; correlation coefficients consistently exceed 0.90.
Comparative Performance
Ranked #21 of 25 clinically evaluated home sleep tests devices

Withings Sleep Analyzer
Withings

Sunrise Home Sleep Test
Sunrise SA

Nox T3s Home Sleep Apnea Test
Nox Medical
You're viewing this
Nox T3s Home Sleep Apnea Test You're viewing this device
Nox Medical
Clinical Context
Our #1-ranked home sleep tests device demonstrated superior performance across key clinical metrics. Compare detailed specifications to make an evidence-based selection.
Clinical Recommendation
Final assessment and prescribing guidance
The Nox T3s is a gold-standard diagnostic tool for clinicians who prioritize data depth. Its blend of calibrated RIP effort and flexible ExG sensors makes it the closest at-home experience to a laboratory study.
Recommend If
You need a medically thorough home sleep study that provides high-fidelity insight into respiratory effort and optional heart or muscle activity.
Avoid If
You are looking for the simplest, least-invasive sensor available and do not require effort belts or ExG data.
Clinical Summary
Composite Score
Recommendation
Where to Buy
Verified purchase options and current availability
CPAP Europa
Complete hardware system purchase for clinical labs; includes 24/7 support.
Medi-Shop.gr
Full Type III HST diagnostic hardware package with AI analysis.
Important Information
- • Prices and availability subject to change
- • Some devices may require a prescription
- • HSA / FSA eligibility: Usually Eligible
- • Purchase from authorized retailers for warranty protection
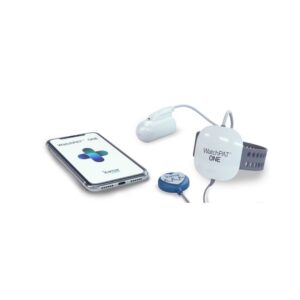
Alternative Devices
Superior diagnostic versatility compared to the WatchPAT One due to ExG channels, though less 'frictionless' for the patient.