
Sonmol Upgraded Digital Peak Flow Meter with ePEF App (SMPF-2S)
Sonmol
by Aluna • Model: K232588
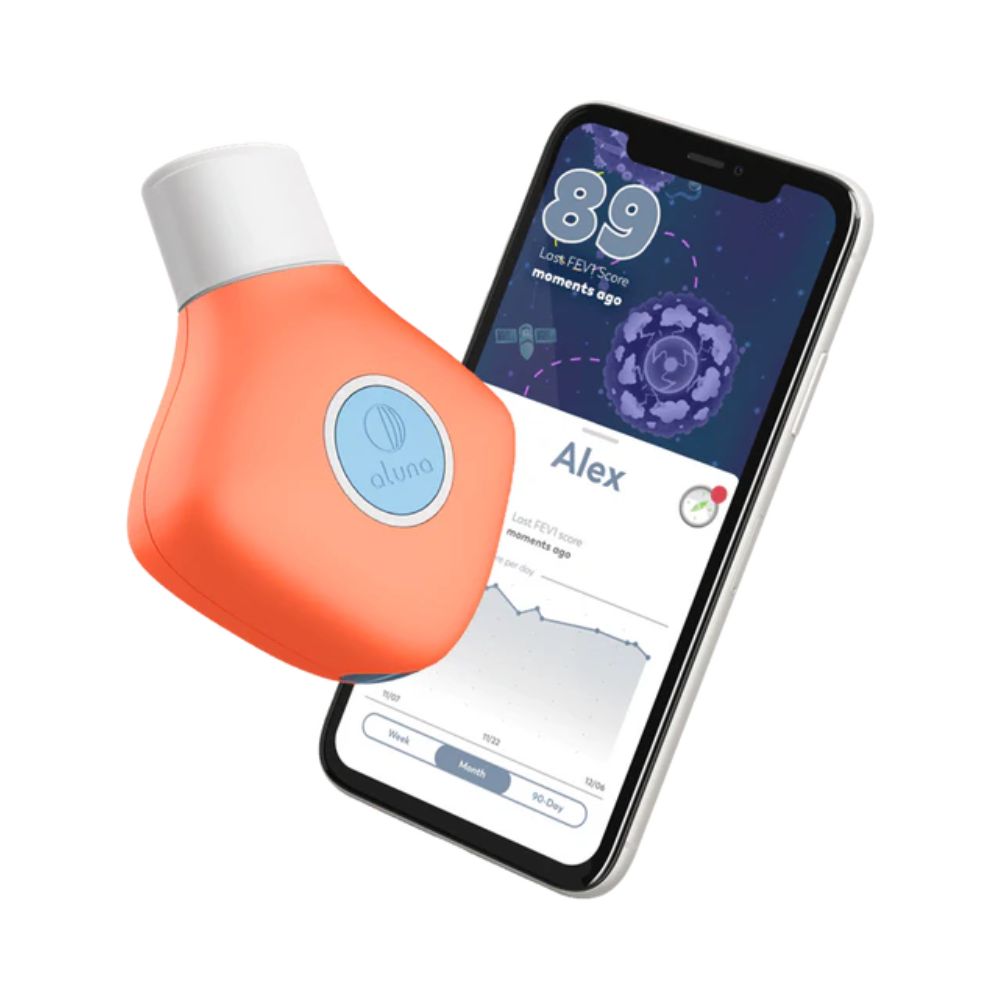
Aluna 2 is a Lilly-type pneumotachometer designed for home monitoring of respiratory health in pediatric and adult users. It utilizes a differential pressure sensor to capture flow data, calculating Forced Expiratory Volume in one second (FEV1) and Peak Expiratory Flow (PEF). The device transmits data via Bluetooth 4.0 LE to an AI-powered mobile app that integrates symptom diaries, air quality tracking, and gamified incentives to improve patient compliance.
Eligibility may vary by provider and plan. Check with your HSA/FSA administrator for specific details.
Evidence-based evaluation by medical professionals
"A highly engaging, hospital-grade smart spirometer that utilizes gamification to encourage daily testing adherence in pediatric and adult patients."
The Aluna 2 is an innovative over-the-counter and prescription smart spirometer intended for monitoring FEV1 (Forced Expiratory Volume in one second) and PEF (Peak Expiratory Flow). Unlike many competitors that use mechanical turbines, the Aluna 2 utilizes a Lilly type pneumotachometer with a differential pressure sensor across a stainless-steel mesh to calculate airflow and volume, offering exceptional durability and hospital-grade accuracy. Cleared by the FDA (K232588) and fully compliant with ATS/ERS 2019 standards, it communicates via Bluetooth 4.0 LE to a dedicated mobile app available on iOS and Android. The standout feature of the Aluna 2 ecosystem is its highly developed companion app, which incorporates engaging breathing games that can be toggled on or off, making it particularly effective for boosting daily adherence in children and teens managing asthma or cystic fibrosis.
Ranked #2 of 14 clinically evaluated home spirometers devices

MIR
Aluna

Aluna
This device scored 1 points behind the highest-rated device in this category. Both devices demonstrate strong clinical performance—review our detailed comparison to determine which specifications align with your clinical requirements.
Verified retailers and current pricing
FSA/HSA eligible; requires prescription for some insurance billing.